
Pollution and biomagnification
The term ‘pollutant’ is a very wide-ranging term. When the introduction or action of something into our environment causes harm it is considered a pollutant.
This could be a harmful chemical such as smoke from a chimney, or it could be a more subtle and transient effect such as floodlights at an evening football match preventing stargazing.
There are many examples of how society has responded to pollution, such as the removal of lead in petrol which affected human health, or the banning of chlorofluorocarbons (CFCs), which damaged the ozone layer.
In both these cases, when the pollution source was removed the levels of them in the environment reduced and consequently so have the effects – albeit with a time delay.
By definition, persistent organic pollutants (POPs) do not break down, so continued introduction of even minute levels of them into an environment leads to accumulation and perhaps magnification of potential harm. PCBs, or polychlorinated biphenyls, are a group of POPs, which while now banned following the Stockholm Agreement in 2001, they were used extensively in electrical and electronic systems up until then. Also the continue to exist by accident as byproducts from manufacturing processes.
PCB levels in Polar Bears can lead to disruption of normal bodily functions, including altering hormone levels. This can the create reproductive issues such as smaller testes size, as well as developmental issues in cubs. Other potential effects include endocrine and immune system disruption, and low bone mineral density.
How do PCBs end up in Polar Bears?
Canadian researchers have found small quantities of POP’s in lichen in the Arctic. The amount of these POP’s increase dramatically in concentration up the Lichen – Caribou (reindeer) – Wolf food chain. Biomagnification in action? This is Trophic transfer: As Caribou eat contaminated lichen, the PCBs in the Lichen are transferred to the Caribou’s fatty tissue. This process repeats as polar bears eat Caribou, with each step of the food chain gaining higher concentrations of the chemicals until they are magnified in Apex predators.

Figure below shows the approximate range of a single female polar bear between 1994 and 1998 (Wiig, 2003). The bear covered an area of around 500,000 km square, in that time, mainly searching for its main prey, seals.
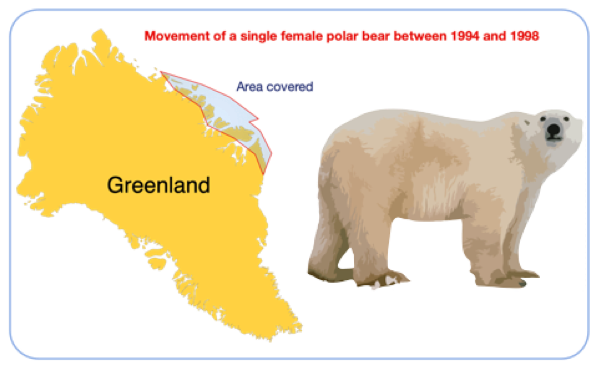
The polar bear was tracked using a satellite tracker. When the is attached the bear has two be drugged. This allows researchers to weigh them and take various samples like fur and teeth for chemical analysis.
While this analysis can tell the researchers much about the health of the polar bear, it is also an important indicator of the effects of pollutants in the system. When the fat of polar bears is analysed it has been found to contain a large amount of certain POPs. Ringed seals have also been found to have high concentrations of POPs in their blubber. Polar bears are at the top of the Arctic food chain, and their diet consists largely of seal blubber, which is very high in fat andPolar bears have a high efficiency for absorbing the fats in their diet. Because PCBs are lipophilic (fat-loving), they are also absorbed along with the fat.
Polars Bears also absorb PCBs and other POPs from fish.
- PCBs in the environment are absorbed by crustaceans in small quantities.
- Fish eat many small crustaceans an and the PCBs absorbed in their biomass are absorbed by the fish and and become bound to their fat stores. The quantity of PCB is magnified.
- Polar bears eat many fish and the PCB transfers from the fish fat stores to Polar bear fat stores.
- The Polar bear has a “magnified” concentration compared to the Fish or the crustaceans.
Even with no industry present, POPs are carried to the artic by winds, ocean currents and rivers. POPs in particular move through air currents from warmer more populated areas to the colder artic, where they fall and settle in the denser air, during snow. Oceanic currents are also responsible for carrying large quantities of POPs from Europe to the artic.
Bioaccumulation of DDT in Peregrin Falcons.
DDT a “good” pollutant?
DDT, Dichlorodiphenyltrichloroethane, was first used as a pesticide widely at the end of the 1940’s. It use was part of the Green Revolution which rsapidly increased global food production after the 1950’s.
However by the 1960’s worries were starting to be raised about the effect of DDT on the environment.

Before World War II the Peregrine falcon and been a common predator of farmland in the West and North of the British at arround 820 breeding pairs. The population was limited only by the number of available nesting sites. Between the 1940’s and 1960’s peregrine numbers declined rapidly in the U.K. and elsewhere in Europe.
Derek Radcliffe (1967) was able to demonstrate that the use of DDT was causing egg thinning and shell breakage. This resulted in fewer young each year and so a smaller population.

figure adapted from:
Banks AN, Coombes RH, Crick, HQP, 2003: The Peregrine Falcon breeding population of the UK & Isle of Man in 2002. Research Report 330. BTO, Thetford https://www.bto.org/sites/default/files/shared_documents/publications/research-reports/2003/rr330.pdf
Ratcliffe, DA, 1967 “Decrease in eggshell weight in certain birds of prey”. Nature. 215 (5097): 208–10
Radcliffe concluded that by eating mice and voles that had eaten grain which had been sprayed with DDT (biomagnification), that the chemical was building up over time – BIOACCUMULATING – in the organs of the peregrines and disrupting the development of egg shells.
We can define bioaccumulation as:
Persistent and non-biodegradable pollutants that can not be broken down (synthesised) can build up in the tissue, organs and cells of an organisms.
After laying their clutch (eggs) when the mother sat on them they would crack as the shells were weakened b the DDT.
DDT created a tipping point (Topic: 1.3)for peregrine populations. A complete ban against the agricultural use of DDT was introduced in the U.K. and many other countries.
Minimata Bay: Biomagnification and Bioaccumulation in Human food chains.
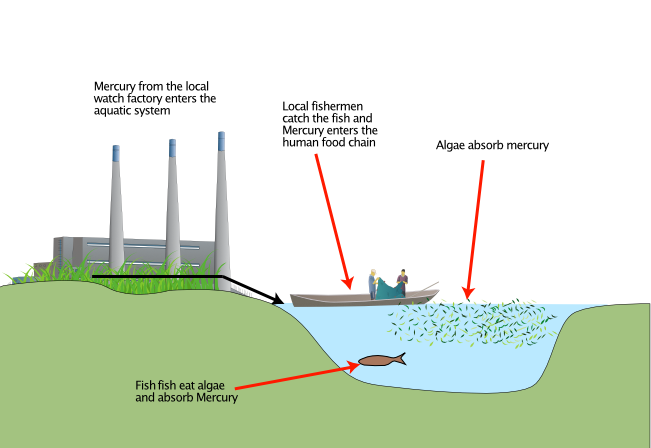
Minamata is a small factory town in Japan, dominated by one factory, The Chisso Factory. Chisso make petrochemical based substances from fertilizer to plastics.
Between 1932 and 1968 Chisso dumped an estimated 27 tons of mercury, used in its production processes into Minamata Bay.
Beginning in the 1950’s, thousands of people started to suffer from mercury poisoning.
Some bacteria (algae) can change mercury to a modified form called methylmercury. Methylmercury is easily absorbed into the bodies of small organisms such as shrimp.
When the shrimp are eaten by fish, the methylmercury enter the fish. The methylmercury does not break down easily and can stay in the fish bodies for a long time. As the fish eat more and more shrimp, the amount of methylmercury increases. The same increase in concentration happens when people then eat the fish. Fish make up a major part of the diet of people around Minamata bay. So biomagnification of the methylmercury occured in the food chain from algae to shrimp, to fish, to humans. But Bioaccumulation also occurred as the concentration in human tissues increased over time.
Work cited:
Ratcliffe, DA, 1967 “Decrease in eggshell weight in certain birds of prey”. Nature. 215 (5097): 208–10
