The Ecosystem
The biogeochemical cycles.
Transfer of matter through the ecosystem is quite different that of energy.
Energy travels from the sun, through food webs and is eventual lost to space as heat.
- Matter is recycled and reused.
- Organisms die are decompose and nutrients (matter) released
- Nutrients may become parts of living things again, when they are taken up by plants
- But they may also be combined into the lithosphere through fossilisation and sedimentation
- Matter may be released to the atmosphere through both natural and anthropogenic processes
These are the BIOGEOCHEMICAL CYCLES
The Biogeochemical cycles ensure chemical elements continue to be available to living organisms. However human impact on these cycles can affect the sustainability of ecosystems.
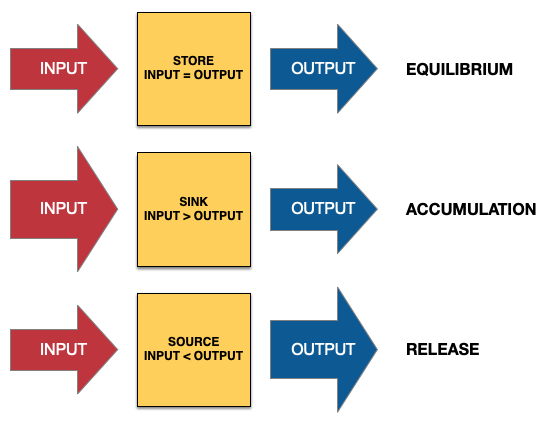
Biogeochemical cycles have stores, sinks and sources.
Stores
- Definition: Also known as reservoirs, these are where elements are held for a period of time, ranging from seconds to millions of years.
- Mechanism: The element is in a state of balance, where the input equals the output. However, they can shift between being a store, sink, or source depending on the net flux.
- Examples:
- Carbon: The atmosphere, oceans, and living organisms like plants and animals.
- Water: Lakes, rivers, and groundwater.
- Nutrients: Soil, biomass, and the atmosphere.
Sinks
- Definition: Areas where an element accumulates over time, with total input exceeding total output.
- Mechanism: The net effect is a long-term removal of the element from the active part of the cycle.
- Examples:
- Carbon: Fossil fuel deposits (like coal and oil), deep ocean, and some rock formations.
- Phosphorus: Sediments and rocks.
Sources
- Definition: Areas where elements are released into the cycle.
- Mechanism: The rate of output is greater than the rate of input.
- Examples:
- Carbon: Volcanic eruptions, burning fossil fuels, respiration, and decomposition.
- Nitrogen: Nitrification in soil, lightning.

The Carbon Cycle
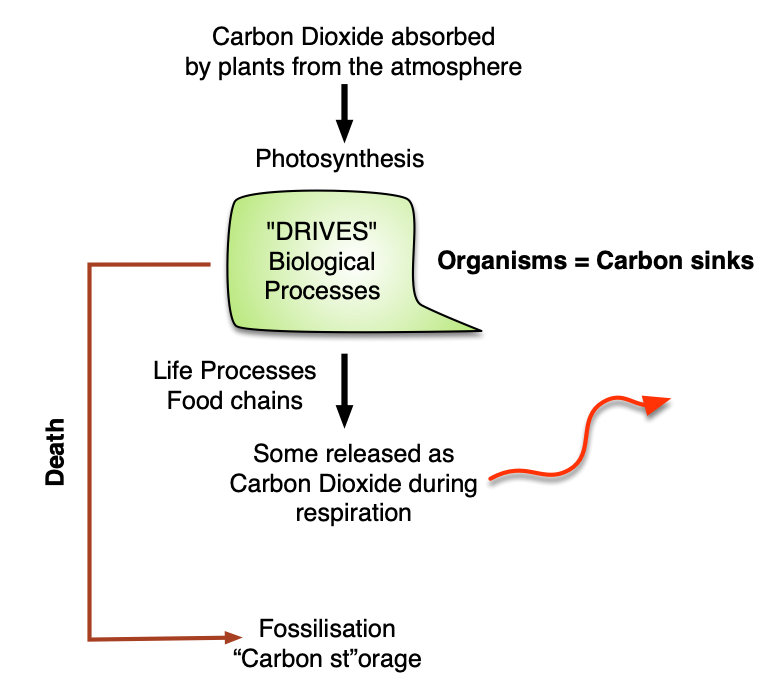
Carbon is an essential element in living systems, providing the chemical framework to form molecules that make up living organisms. Carbon makes up around 0.03% of the atmosphere as carbon dioxide, and is present in the Oceans as carbonate and bicarbonates and in rocks such as limestone and coal.
Carbon cycles between living (biotic) and non-living (abiotic) chemical cycles: carbon is fixed by photosynthesis and released back to the atmosphere through respiration. Carbon is also released back to the atmosphere through combustion, including fossil fuels and biomass.
Carbon can remain locked in either cycle for long periods of time. ie in the wood of trees or as coal and oil.
Human activity has disrupted the balance of the global carbon cycle (carbon budget) through increased combustion, land use changes and deforestation.
Organisms, crude oil and natural gas contain organic stores of organic carbon. The atmosphere, soils and oceans store inorganic carbon. The amount of time Carbon (or any molecule in the biogeochemical cycles) is held in storage, is called residence time. Humans have disrupted the residence time of geologically stored carbon through mining and extraction of fossil fuels: Coal, oil and gas. Without human interference the residence time would by hundreds of millions of years.
Carbon Sequestration

Environmental systems and societies guide
Carbon sequestration is the process of capturing gaseous and atmospheric carbon dioxide and storing it in a solid or liquid form. Trees and other plants sequester carbon naturally by absorbing carbon dioxide and converting it into biomass. Organic matter is fossilised into coal, oil and natural gas.
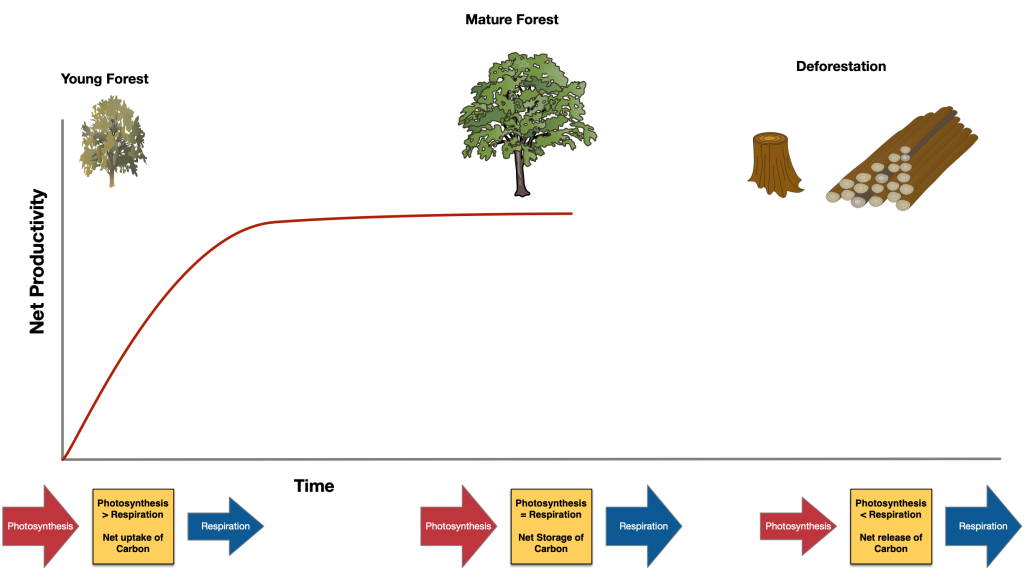
In an ecosystem, the difference between total inputs and outputs is the net accumulation or release of carbon. If photosynthesis exceeds cellular respiration in an ecosystem there is a net uptake of carbon dioxide, and if cellular respiration exceeds photosynthesis there is a net release of carbon dioxide. In a young forest, the trees act as a sink as net productivity is high, a mature forest acting as a store when photosynthesis equals respiration and when a forest is logged (deforestation) or following a forest fire carbon is released to the atmosphere and the ecosystem becomes a carbon source.
Agricultural systems can act as carbon stores, sources and sinks, depending on the techniques used. Some agricultural methods, such as crop rotation, cover crops and no till, will promote the role, of soil as a carbon sink whereas drainage of wetland, monoculture and heavy tillage will promote the role of soil as a carbon source. Cropping over a longer timescale such as in forestry for timber production and the subsequent use of harvested products will also affect these roles (illustrated above).



Fossil Fuels and the Carbon Cycle
Fossil fuels were plant and animal matter laid down between sediments millions of years ago and through fossilisation become Coal, Oil and Natural gas – a period when ecosystems acted as net sinks of carbon. Geologically they have a very residence time. However when we burn them as fuel they become a carbon source.


Oceans as Carbon Sinks and Sources
Carbon dioxide is absorbed into the oceans by dissolving and is released as a gas when it comes out of a solution. While oceans act as a carbon sink, the human use of fossil fuels releases inorganic carbon at a faster rate than oceans can absorb it. Environmental systems and societies guide. Increases in concentrations of dissolved carbon dioxide cause ocean acidification, harming marine animals even small decreases in pH can interfere with calcium carbonate deposition in mollusc shells and 2.1 coral skeletons.
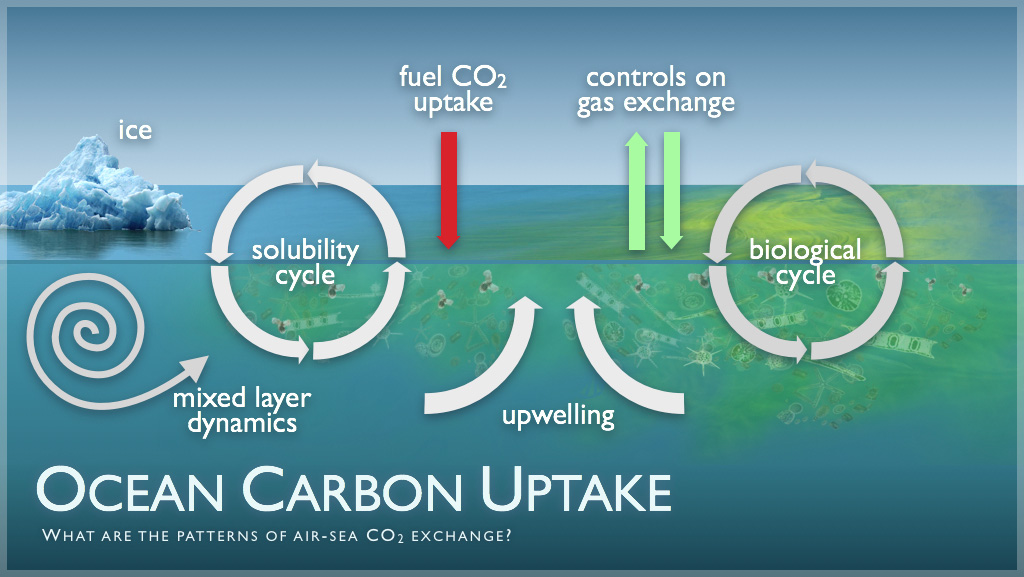
Open the link for a more detailed explanation of how the cceans act as a carbon sink, but also how man’s activities create a tension with this role.

